A simple mathematical approach to identifying baseline predictors of the effectiveness of carbonic anhydrase inhibitors in intraocular pressure reduction
Main Article Content
Abstract
Purpose
To identify mathematically the baseline physiological predictors of the intraocular pressure (IOP)-lowering effectiveness of topical carbonic anhydrase inhibitors (CAIs).
Materials and Methods
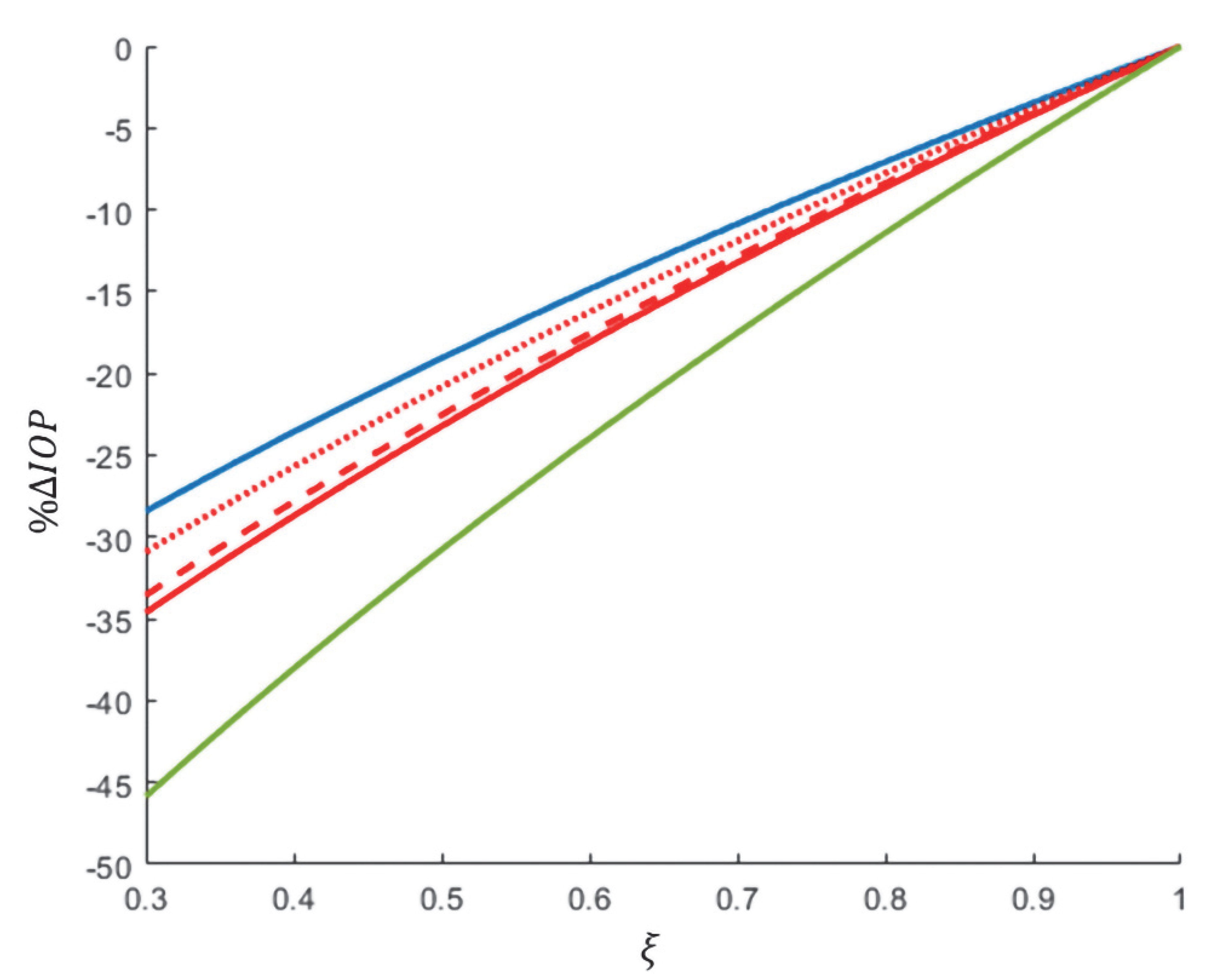
Assuming a steady state of aqueous humor flow, mathematical equations describing IOP were formulated by incorporating related physiological variables. Global sensitivity analysis was then performed, in which all variables were varied simultaneously across their clinically reported ranges. This computational approach quantified the percentage reduction in IOP achieved by CAIs and identified the key determinants underlying variability in their effectiveness.
Results
A reduction in the solute influx via active transport by the ciliary epithelium, mediated by CAIs, was positively associated with the magnitude of IOP reduction. Among the physiological parameters in this model—trabecular outflow facility, uveoscleral outflow fraction, episcleral venous pressure, mean arterial pressure, plasma protein concentration, albumin/globulin ratio, and plasma osmolarity—a higher baseline trabecular outflow facility was the strongest predictor of reduced effectiveness of CAIs, in terms of the percentage of IOP reduction. Other baseline physiological parameters had no significant effect on the effectiveness of CAIs.
Conclusions
Certain patient characteristics may be associated with compromised effectiveness of CAIs in terms of percentage IOP reduction. These characteristics, potentially associated with higher trabecular outflow facility, may include a lower baseline IOP or prior use of other trabecular outflow-enhancing medications before CAI initiation. Further studies are required to validate the clinical significance of these findings.
Downloads
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
Pazos M, Traverso CE, Viswanathan A, et al. European Glaucoma Society – Terminology and guidelines for glaucoma, 6th ed. Br J Ophthalmol 2025;109(Suppl 1):1-212.
Stoner A, Harris A, Oddone F, et al. Topical carbonic anhydrase inhibitors and glaucoma in 2021: where do we stand? Br J Ophthalmol 2022;106:1332-7.
Sugrue MF. Pharmacological and ocular hypotensive properties of topical carbonic anhydrase inhibitors. Prog Retin Eye Res 2000;19:87-112.
Boyer S, Gay D. Additive effect of dorzolamide hydrochloride to patients taking travoprost: a retrospective study. Optometry 2008;79:501-4.
Holló G, Chiselita D, Petkova N, et al. The efficacy and safety of timolol maleate versus brinzolamide each given twice daily added to travoprost in patients with ocular hypertension or primary open-angle glaucoma. Eur J Ophthalmol 2006;16:816-23.
Li T, Lindsley K, Rouse B, et al. Comparative effectiveness of first-line medications for primary open-angle glaucoma: a systematic review and network meta-analysis. Ophthalmology 2016;123:129-40.
Michaud JE, Friren B. Comparison of topical brinzolamide 1% and dorzolamide 2% eye drops given twice daily in addition to timolol 0.5% in patients with primary open-angle glaucoma or ocular hypertension. Am J Ophthalmol 2001;132:235-43.
Oztürk F, Ermiş SS, Inan UU, et al. Comparison of the efficacy and safety of dorzolamide 2% when added to brimonidine 0.2% or timolol maleate 0.5% in patients with primary open-angle glaucoma. J Ocul Pharmacol Ther 2005;21:68-74.
Pfeiffer N; Travatan Adjunctive Treatment Study Group. Timolol versus brinzolamide added to travoprost in glaucoma or ocular hypertension. Graefes Arch Clin Exp Ophthalmol 2011;249:1065-71.
Reis R, Queiroz CF, Santos LC, et al. Randomized, investigator-masked, 4-week study comparing timolol maleate 0.5%, brinzolamide 1%, and brimonidine tartrate 0.2% as adjunctive therapies to travoprost 0.004% in adults with primary open-angle glaucoma or ocular hypertension. Clin Ther 2006;28:552-9.
Lyubimov G, Moiseeva I, Stein A. Dynamics of the intraocular fluid: mathematical model and its main consequences. Fluid Dynamics 2007;42:684-94.
Dendumrongsup W. Identifying baseline predictors of selective laser trabeculoplasty effectiveness: an alternative mathematical approach. Cureus 2024;16:e54116.
Nitta S, Ohnuki T, Ohkuda K, et al. The corrected protein equation to estimate plasma colloid osmotic pressure and its development on a nomogram. Tohoku J Exp Med 1981;135:43-9.
Feitl ME, Krupin T. Carbonic Anhydrase Inhibitors in Ophthalmology. In: Dodgson SJ, Tashian RE, Gros G, Carter ND, eds. The Carbonic Anhydrases: Cellular Physiology and Molecular Genetics. Boston, MA: Springer US; 1991:163-70.
Schmidl D, Schmetterer L, Garhöfer G, Popa-Cherecheanu A. Pharmacotherapy of glaucoma. J Ocul Pharmacol Ther 2015;31:63-77.
Grant WM. Clinical measurements of aqueous outflow. AMA Arch Ophthalmol 1951;46:113-31.
Sacco R, Guidoboni G, Jerome JW, et al. A theoretical approach for the electrochemical characterization of ciliary epithelium. Life 2020;10:8.
Dvoriashyna M, Foss AJE, Gaffney EA, Repetto R. A mathematical model of aqueous humor production and composition. Invest Ophthalmol Vis Sci 2022;63:1.
Larsson L-I, Rettig ES, Brubaker RF. Aqueous flow in open-angle glaucoma. Arch Ophthalmol 1995;113:283-6.
Alm A, Nilsson SFE. Uveoscleral outflow—a review. Exp Eye Res 2009;88:760-8.
Sit AJ, McLaren JW. Measurement of episcleral venous pressure. Exp Eye Res 2011;93:291-8.
Kiers D, Hofstra J, Wetzels J. Oscillometric blood pressure measurements: differences between measured and calculated mean arterial pressure. Neth J Med 2008;66:474-9.
Busher JT. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd ed. Boston: Butterworths; 1990.
Smith GS, Walter GL, Walker RM. Clinical pathology in non-clinical toxicology testing. In: Haschek WM, Rousseaux CG, Wallig MA, eds. Haschek and Rousseaux’s Handbook of Toxicologic Pathology (Third Edition). Boston: Academic Press; 2013:565-94.
Rasouli M. Basic concepts and practical equations on osmolality: biochemical approach. Clin Biochem 2016;49:936-41.
Johnson M, McLaren JW, Overby DR. Unconventional aqueous humor outflow: a review. Exp Eye Res 2017;158:94-111.
Brubaker RF. Targeting outflow facility in glaucoma management. Surv Ophthalmol 2003;48 Suppl 1:S17-20.
Brubaker RF. Mechanism of action of bimatoprost (Lumigan). Surv Ophthalmol 2001;45 Suppl 4:S347-51.
Overby DR, Bertrand J, Schicht M, et al. The structure of the trabecular meshwork, its connections to the ciliary muscle, and the effect of pilocarpine on outflow facility in mice. Invest Ophthalmol Vis Sci 2014;55:3727-36.
Stamer WD, Acott TS. Current understanding of conventional outflow dysfunction in glaucoma. Curr Opin Ophthalmol 2012;23:135-43.
Inoue T, Tanihara H. Rho-associated kinase inhibitors: a novel glaucoma therapy. Prog Retin Eye Res 2013;37:1-12.
Kazemi A, McLaren JW, Trese MGJ, et al. Effect of timolol on aqueous humor outflow facility in healthy human eyes. Am J Ophthalmol 2019;202:126-32.
Webers C, Valk R, Schouten J, et al. Intraocular pressure-lowering effect of adding dorzolamide or latanoprost to timolol. a meta-analysis of randomized clinical trials. Ophthalmology 2007;114:40-6.
O’Connor DJ, Martone JF, Mead A. Additive intraocular pressure lowering effect of various medications with latanoprost. Am J Ophthalmol 2002;133:836-7.