A retrospective study on the outcomes of Ahmed valve versus Ahmed valve combined with fluocinolone implant in uveitic glaucoma
Main Article Content
Abstract
Purpose
To compare the intraocular pressure (IOP) outcomes of Ahmed glaucoma valve (AGV) surgery alone versus AGV with fluocinolone implant in uveitic glaucoma patients.
Methods
We identified uveitic glaucoma patients with AGV surgery alone and AGV surgery combined with fluocinolone implant. Demographic information, visual acuity, and IOP were recorded at preoperative visits and 1, 6, and 12 months after surgery. Incidence of hypertensive phase, defined as an IOP of >21 mm Hg or use of additional treatment to lower IOP occurring any time between 7 days to 6 months postoperatively, was investigated. Multilevel mixed effects models were performed.
Results
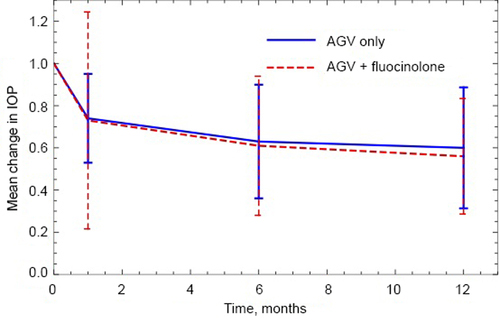
Eighteen eyes of 13 uveitic glaucoma patients with 1-year follow-up data were included. There were 11 eyes of 9 patients (mean age, 56.5 years; 63.6% male) in the AGV group and 7 eyes of 4 patients (mean age, 61.3 years; 71.4% male) in the AGV + fluocinolone group. There was no significant difference in visual acuity change at 1 year after surgery between groups, although visual acuity improvement was significant in the AGV group (P = 0.01). The hypertensive phase occurred in 91% of AGV patients and 43% of AGV + fluocinolone patients (P = 0.30), with onset of 8-40 days (mean, 18 days) after surgery. IOP and number of glaucoma medications decreased at the 1-year postoperative visits in both the AGV group (P < 0.0001, P < 0.0001) and the AGV + fluocinolone group (P = 0.001, P < 0.0001). Compared to the AGV group, the AGV + fluocinolone group used fewer glaucoma medications (0.28 vs 1.30 [P = 0.01]) and had better inflammation control (P = 0.02). The surgical complication rates were similar between groups.
Conclusions
In uveitic glaucoma, AGV with fluocinolone achieves a similar, desired IOP control but with fewer glaucoma medications than AGV alone.
Downloads
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.