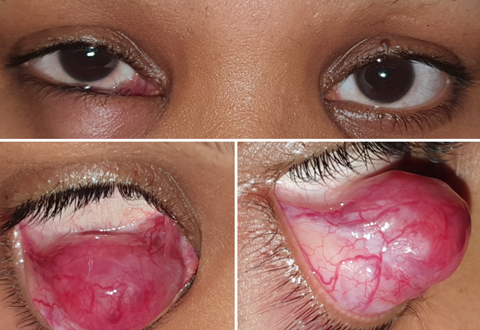
A 29-year-old woman with right lower lid mass
Main Article Content
Abstract
Extraosseous Ewing sarcoma is a rare entity mostly seen in the extremities and chest cavity in older individuals. Although osseous orbital Ewing sarcoma has been reported, extraosseous orbital involvement is extremely rare. We report the case of a young woman who presented with lower eyelid mass lesion. The results of histopathological and immunohistochemical analysis were suggestive of Ewing sarcoma. Radiological imaging and a PET-CT ruled out any bony orbital or systemic involvement. The patient was started on a standard chemotherapeutic protocol for Ewing sarcoma.
Downloads
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
Hornicek FJ, Elizabeth HB. Clinical presentation, staging, and prognostic factors of Ewing sarcoma. In: UpToDate, Pollock RE, Pappo AS, Maki R (Ed), UpToDate, Waltham, MA.2020. Accessed October 22, 2023.
Kaliki S, Rathi SG, Palkonda VAR. Primary orbital Ewing sarcoma family of tumors: a study of 12 cases. Eye (Lond) 2018;32:615-21. DOI: https://doi.org/10.1038/eye.2017.278
Alio JL 2nd, Sales-Sanzv, Vaz MA, et al. Primary extraosseous Ewing sarcoma of the orbit. Ophthalmic Plast Reconstr Surg 2013;29:91-3. DOI: https://doi.org/10.1097/IOP.0b013e3182771549
Lane KA, Katowitz JA. Ewing sarcoma presenting as a subconjunctival mass. Ophthal Plast Reconstr Surg 2009;25:61-3. DOI: https://doi.org/10.1097/IOP.0b013e31819361fd
Li T, Goldberg RA, Becker B, et al. Primary orbital extraskeletal Ewing sarcoma. Arch Ophthalmol 2003;121:1049-52. DOI: https://doi.org/10.1001/archopht.121.7.1049
Alexander A, Hunter K, Rubin M, et al. Extraosseous Ewing’s sarcoma: pictorial review of imaging findings, differential diagnosis, and pathologic correlation. Indian J Radiol Imaging 2021;31:203-9. DOI: https://doi.org/10.1055/s-0041-1729770
Alhejaily AG, Yassin EM. Genetic alterations in Ewing’s sarcoma: what do we know so far? Musculoskelet Surg Res 2022;6:117-20. DOI: https://doi.org/10.25259/JMSR_148_2021
McCuiston A, Bishop JA. Usefulness of NKX2.2 immunohistochemistry for distinguishing Ewing sarcoma from other sinonasal small round blue cell tumors. Head Neck Pathol 2018;12:89-94. DOI: https://doi.org/10.1007/s12105-017-0830-1
Gebhardt MC, DuBois SG. Treatment of Ewing sarcoma. In: UpToDate, Pollock RE, Pappo AS, Maki R (Ed), UpToDate, Waltham, MA. (Accessed on October 22, 2023)
Craft A, Cotterill S, Malcolm A, et al. Ifosfamide-containing chemotherapy in Ewing’s sarcoma: the Second United Kingdom Children’s Cancer Study Group and the Medical Research Council Ewing’s Tumor Study. J Clin Oncol 1998;16:3628-33. DOI: https://doi.org/10.1200/JCO.1998.16.11.3628
Paulussen M, Ahrens S, Dunst J, et al. Localized Ewing tumor of bone: final results of the cooperative Ewing’s Sarcoma Study CESS 86. J Clin Oncol 2001;19:1818-29. DOI: https://doi.org/10.1200/JCO.2001.19.6.1818
Ahmed SK, Robinson SI, Okuno SH, et al. Adult Ewing sarcoma: survival and local control outcomes in 36 patients with metastatic disease. Am J Clin Oncol 2014;37:423-9. DOI: https://doi.org/10.1097/COC.0b013e31827de65e
Ahmad R, Mayol BR, Davis M, et al. Extraskeletal Ewing’s sarcoma. Cancer 1999;85:725-31. DOI: https://doi.org/10.1002/(SICI)1097-0142(19990201)85:3<725::AID-CNCR23>3.0.CO;2-2
Castex MP, Rubie H, Stevens MC, et al. Extraosseous localized Ewing tumors: improved outcome with anthracyclines—the French Society of Pediatric Oncology and International Society of Pediatric Oncology. J Clin Oncol 2007;25:1176-82. DOI: https://doi.org/10.1200/JCO.2005.05.0559
Brennan B, Kirton L, Marec-Bérard P, et al. Comparison of two chemotherapy regimens in patients with newly diagnosed Ewing sarcoma (EE2012): an open-label, randomised, phase 3 trial. Lancet 2022;400:1513-21. DOI: https://doi.org/10.1016/S0140-6736(22)01790-1